As the core location for drug production, pharmaceutical workshops have extremely stringent requirements for air quality, cleanliness, and safety. The choice of building materials directly determines drug quality and production safety. Mechanism sulfur oxygen magnesium cleanroom panels, with their unique performance advantages, have become the preferred building material for pharmaceutical workshops. Combining industry experience and practical application scenarios, this article provides a detailed analysis of its core advantages, application scenarios, and usage precautions, offering practical reference for pharmaceutical companies.

The core reason why mechanized magnesium oxysulfate purification panels have become the first choice for pharmaceutical workshops lies in their three key performance characteristics, precisely addressing industry pain points and comprehensively meeting production requirements.
Firstly, superior purification capabilities build a solid aseptic defense. Pharmaceutical production has extremely high requirements for a sterile environment. The smooth, non-porous surface of this panel effectively reduces dust accumulation; its special formula achieves anti-static effects, preventing electrostatic adsorption of particulate matter that contaminates drugs; and it also possesses excellent antibacterial properties, inhibiting the growth of harmful microorganisms such as Escherichia coli and Staphylococcus aureus, ensuring a clean and sterile production environment from the source.
Secondly, excellent physical properties ensure production safety. Pharmaceutical workshops often involve complex scenarios such as high-temperature sterilization and equipment operation. This type of board is heat-resistant, high-strength, and not easily deformed or damaged. It meets the Class A non-combustible fire standard, effectively preventing the spread of fire and producing no toxic or harmful gases during combustion, significantly improving workshop safety and complying with industry fire safety regulations.
Thirdly, its energy-saving and environmentally friendly characteristics reduce operating costs. The board is produced using environmentally friendly raw materials and processes, with no harmful gas emissions, conforming to the concept of green production. At the same time, its excellent thermal insulation performance and low thermal conductivity reduce workshop energy consumption, helping companies achieve energy conservation and improved operational efficiency.
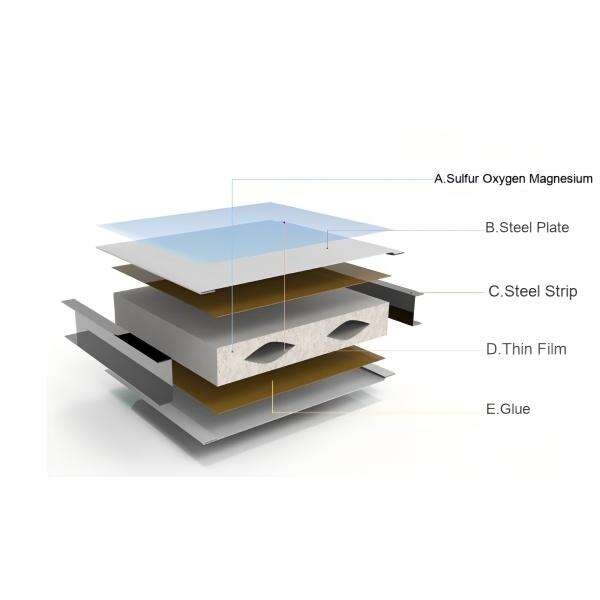
Based on the above advantages, the mechanically produced magnesium oxysulfate purification board is widely used in various core scenarios of pharmaceutical workshops, spanning the entire production process.
As a primary building material for walls and ceilings, its strong sealing and high cleanliness effectively reduce airborne particulate pollution, preventing dust from falling onto medicines or production equipment, directly ensuring drug quality. As functional area partitions and doors, these panels, with their tight sealing performance, effectively block airflow between different areas, preventing cross-contamination by microorganisms and particles, thus meeting the requirements of workshop zoning management. Furthermore, they can be used to manufacture clean benches and sterilization equipment enclosures, further enhancing the local sterile environment and providing added protection for critical production processes.
High-quality panels require standardized use and maintenance to fully realize their advantages and extend their service life. Considering the special environment of pharmaceutical workshops, three key points need attention:
Installation and Construction:Strictly control cleanliness and sealing standards during installation. Clean the site of dust and debris before construction. Follow cleanroom engineering specifications during installation. Use specialized sealant at joints. Use cleanroom-specific accessories for fasteners. It is recommended that this be done by a professional team.
Maintenance and Care:Follow gentle cleaning principles. Use a neutral, non-corrosive cleaner with a soft cloth to wipe, avoiding damage to the panel coating. Keep the workshop well-ventilated and dry. Regularly inspect and promptly repair any damage to high-frequency contact areas.
Safety Monitoring:Conduct regular safety monitoring, focusing on fire resistance, purification, and sealing indicators. Record monitoring data to ensure compliance with industry standards.

In summary, with its advantages in purification, safety, and environmental protection, it perfectly meets the needs of pharmaceutical workshops. Standardized installation, scientific maintenance, and regular monitoring are key to realizing its value. Glostar, with its deep expertise in purification materials, custom-produces mechanical magnesium oxysulfate purification panels that meet pharmaceutical standards. We provide comprehensive services from selection and production to installation and after-sales support, helping companies build safe, clean, and compliant production environments. Customization inquiries are welcome.
 Hot News
Hot News2026-05-11
2026-05-08
2026-05-07
2026-05-06
2026-04-30
2026-04-27